Chitosan-coated CMC and carbopol hydrogel beads for controlled release of metformin in diabetes management
DOI:
https://doi.org/10.69857/joapr.v13i2.1006Keywords:
Metformin HCl, beads, Diabetes Mellitus, chitosan, carboxymethyl cellulose sodium, carbopolAbstract
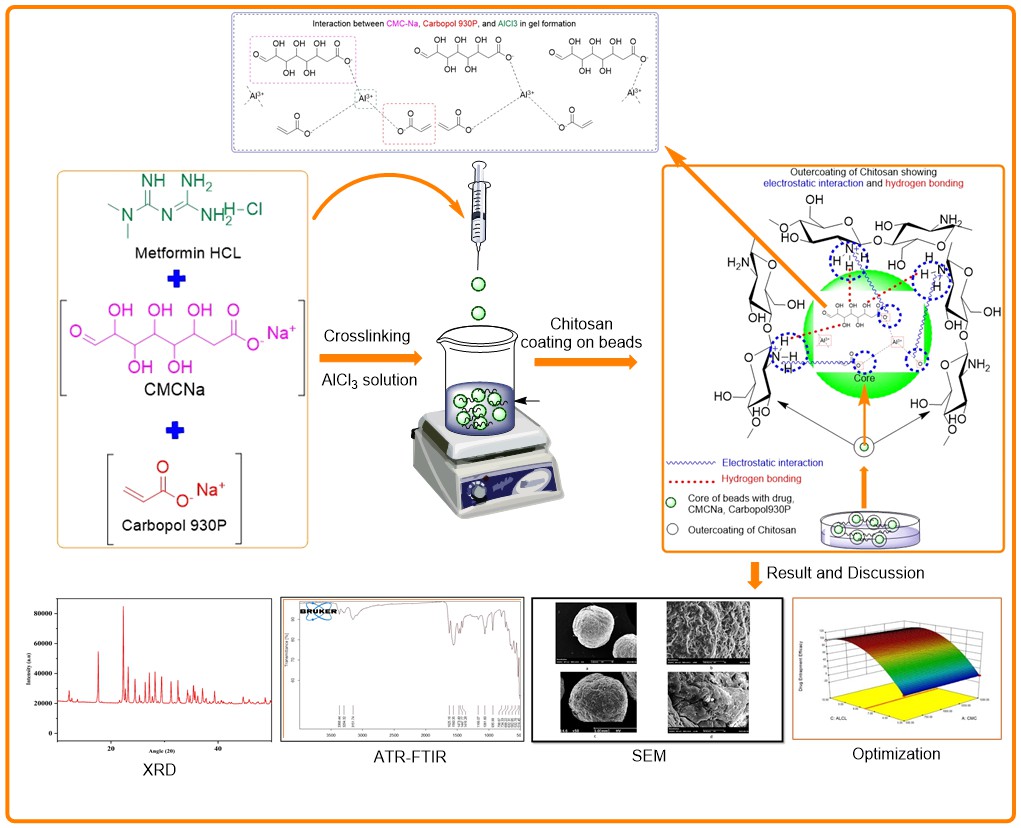
Background: Current research aims to fabricate carboxymethyl cellulose sodium (CMC-Na) and carbopol hydrogel beads. Gleichzeitig, beads were coated with chitosan to enhance the controlled release of the drug Metformin HCl (MET), which serves as a model drug for diabetes mellitus (DM). Methodology: The MET beads were synthesized through the ionotropic gelation process. The foundation of ionotropic gelation is a polyelectrolyte’s capacity to cross-link to create hydrogels when counterions are present. The negatively charged carboxylate groups (-COO⁻) on CMC-Na form electrostatic interactions with the positively charged aluminium ions (Al³⁺) from AlCl3. The quality-by-design approach was employed to optimize process factors in preparing hydrogel beads. A comprehensive evaluation of the beads covered various aspects such as particle size, scanning electron microscopy, percentage yield, Fourier transform infrared spectroscopy, X-ray diffraction, entrapment efficiency (EE), and in vitro drug release. Results and Discussion: The beads were spherical, with an average particle diameter of 153.6 to 231.5 μm. The entrapment efficiency percentage range is 94.4% and 97.83% for MET-loaded and chitosan-coated MET-loaded beads, respectively. Therefore, in-vitro drug release of the optimized MET-loaded beads is 55.5 %, and chitosan-coated MET-loaded beads are approximately 48.8% achieved in 10 hours. Conclusion: Chitosan-coated CMC-Na and carbopol hydrogel beads showed good MET encapsulation and sustained release, improving structural integrity and drug release. The ionotropic gelation process created stable, homogeneous beads, making this delivery method viable for oral sustained-release MET formulations.
Downloads
References
Tentolouris A, Vlachakis P, Tzeravini E, Eleftheriadou I, Tentolouris N. SGLT2 inhibitors: A review of their antidiabetic and cardioprotective effects. Int J Environ Res Public Health, 16, 1–27 (2019) https://doi.org/10.3390/ijerph16162965.
M A, Gogate PR. Improved synthesis of metformin hydrochloride-sodium alginate (MH-NaALG) microspheres using ultrasonic spray drying. Heliyon, 10, e28205 (2024) https://doi.org/10.1016/j.heliyon.2024.e28205.
Kirpichnikov D, McFarlane SI, Sowers JR. Metformin: An Update. Ann Intern Med, 137, 25–33 (2002) https://doi.org/10.7326/0003-4819-137-1-200207020-00009.
Kurdi A, Alhussaini W, Alawaji A, Alhudathi A, Alharbi R, Binsaleh F, Alghamidi Y, Al Bekairy A, Alkatheri A, Islam I, Farh I, Ghanem E, Mansour M. Comparative performance of liquid chromatography and spectrophotometry in determining metformin hydrochloride within pharmaceutical formulations. Heliyon, 10, e32551 (2024) https://doi.org/10.1016/j.heliyon.2024.e32551.
Ozoude CH, Azubuike CP, Ologunagba MO, Tonuewa SS, Igwilo CI. Formulation and development of metformin-loaded microspheres using Khaya senegalensis (Meliaceae) gum as co-polymer. Futur J Pharm Sci, 6, (2020) https://doi.org/10.1186/s43094-020-00139-6.
Karzar Jeddi M, Mahkam M. Magnetic nano carboxymethyl cellulose-alginate/chitosan hydrogel beads as biodegradable devices for controlled drug delivery. Int J Biol Macromol, 135, 829–38 (2019) https://doi.org/10.1016/j.ijbiomac.2019.05.210.
Hosny EA, Al-Helw AA-RM. Effect of coating of aluminum carboxymethylcellulose beads on the release and bioavailability of diclofenac sodium. Pharm Acta Helv, 72, 255–61 (1998) https://doi.org/10.1016/S0031-6865(97)00040-X.
Lopez CG, Colby RH, Cabral JT. Electrostatic and Hydrophobic Interactions in NaCMC Aqueous Solutions: Effect of Degree of Substitution. Macromolecules, 51, 3165–75 (2018) https://doi.org/10.1021/acs.macromol.8b00178.
Zakzak K, Semenescu AD, Moacă EA, Predescu I, Drăghici G, Vlaia L, Vlaia V, Borcan F, Dehelean CA. Comprehensive Biosafety Profile of Carbomer-Based Hydrogel Formulations Incorporating Phosphorus Derivatives. Gels, 10, (2024) https://doi.org/10.3390/gels10070477.
López-Cacho JM, González-R PL, Talero B, Rabasco AM, González-Rodrguez ML. Robust optimization of alginate-Carbopol 940 bead formulations. The Scientific World Journal, 2012, (2012) https://doi.org/10.1100/2012/605610.
Mahmood A, Mahmood A, Ibrahim MA, Hussain Z, Ashraf MU, Salem-Bekhit MM, Elbagory I. Development and Evaluation of Sodium Alginate/Carbopol 934P-Co-Poly (Methacrylate) Hydrogels for Localized Drug Delivery. Polymers (Basel), 15, (2023) https://doi.org/10.3390/polym15020311.
de Souza HKS, Bai G, Gonçalves M do P, Bastos M. Whey protein isolate–chitosan interactions: A calorimetric and spectroscopy study. Thermochim Acta, 495, 108–14 (2009) https://doi.org/10.1016/j.tca.2009.06.008.
Zhao D, Shen X. Preparation of Chitosan-Diatomite/Calcium Alginate Composite Hydrogel Beads for the Adsorption of Congo Red Dye. Water (Switzerland), 15, (2023) https://doi.org/10.3390/w15122254.
Fatullayeva S, Tagiyev D, Zeynalov N, Mammadova S, Aliyeva E. Recent advances of chitosan-based polymers in biomedical applications and environmental protection. Journal of Polymer Research, 29, 259 (2022) https://doi.org/10.1007/s10965-022-03121-3.
Zia Q, Tabassum M, Gong H, Li J. A Review on Chitosan for the Removal of Heavy Metals Ions. Journal of Fiber Bioengineering and Informatics, 12, 103–28 (2019) https://doi.org/10.3993/jfbim00301.
Essifi K, Lakrat M, Berraaouan D, Fauconnier ML, El Bachiri A, Tahani A. Optimization of gallic acid encapsulation in calcium alginate microbeads using Box-Behnken Experimental Design. Polymer Bulletin, 78, 5789–814 (2021) https://doi.org/10.1007/s00289-020-03397-9.
Gadziński P, Froelich A, Jadach B, Wojtyłko M, Tatarek A, Białek A, Krysztofiak J, Gackowski M, Otto F, Osmałek T. Ionotropic Gelation and Chemical Crosslinking as Methods for Fabrication of Modified-Release Gellan Gum-Based Drug Delivery Systems. Pharmaceutics, 15, 108 (2022) https://doi.org/10.3390/pharmaceutics15010108.
Hu Y, Hu S, Zhang S, Dong S, Hu J, Kang L, Yang X. A double-layer hydrogel based on alginate-carboxymethyl cellulose and synthetic polymer as sustained drug delivery system. Sci Rep, 11, 1–14 (2021) https://doi.org/10.1038/s41598-021-88503-1.
Komati S, Swain S, Rao MEB, Jena BR, Dasi V. Mucoadhesive multiparticulate drug delivery systems: An extensive review of patents. Adv Pharm Bull, 9, 521–38 (2019) https://doi.org/10.15171/apb.2019.062.
Nualkaekul S, Lenton D, Cook MT, Khutoryanskiy V V., Charalampopoulos D. Chitosan coated alginate beads for the survival of microencapsulated Lactobacillus plantarum in pomegranate juice. Carbohydr Polym, 90, 1281–7 (2012) https://doi.org/10.1016/j.carbpol.2012.06.073.
Deepak P, Kumar P, Arya DK, Pandey P, Kumar S, Parida BP, Narayan G, Singh S, Rajinikanth PS. c(RGDfK) anchored surface manipulated liposome for tumor-targeted tyrosine kinase inhibitor (TKI) delivery to potentiate liver anticancer activity. Int J Pharm, 642, 123160 (2023) https://doi.org/10.1016/j.ijpharm.2023.123160.
Aggarwal G, Nagpal M. Pharmaceutical Polymer Gels in Drug Delivery. Polymer Gels: Perspectives and Applications. (Thakur Vijay Kumar and Thakur Manju Kumari and Voicu Stefan Ioan ed.), Springer Singapore, Singapore, pp. 249–84 (2018) https://doi.org/10.1007/978-981-10-6080-9_10
Altam AA, Zhu L, Huang W, Huang H, Yang S. Polyelectrolyte complex beads of carboxymethylcellulose and chitosan: The controlled formation and improved properties. Carbohydrate Polymer Technologies and Applications, 2, 100100 (2021) https://doi.org/10.1016/j.carpta.2021.100100.
Sabbagh BA, Kumar PV, Chew YL, Chin JH, Akowuah GA. Determination of metformin in fixed-dose combination tablets by ATR-FTIR spectroscopy. Chemical Data Collections, 39, 100868 (2022) https://doi.org/10.1016/j.cdc.2022.100868.
Published
How to Cite
Issue
Section
Copyright (c) 2025 Sachin Gupta, Swati Dubey, Sanjeev Kumar Patel, Anshu Priyanka Lakra, Sunita Minz

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
















